Process
The Reverse Osmosis System
Osmosis is a natural process that occurs when two volumes of water are separated by a semi-permeable membrane, which has holes that are small enough to trap contaminants, but allows water to flow through.
Water will flow through the semi-permeable membrane from the side of the low solute concentration (fewer contaminants) to the side of the high solute concentration (more contaminants) in order to restore equilibrium between the two sides. This flow of water may be stopped or even reversed if external pressure is applied to the higher solute concentration, ensuring the purification of the water.
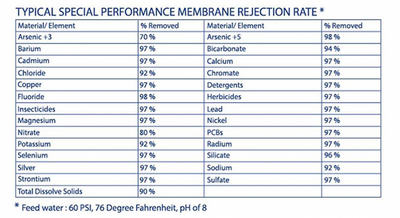
The table on the right demonstrates the different types of contaminants that are removed from the water through our system.